Once-daily dosing1

Once-daily dosing1
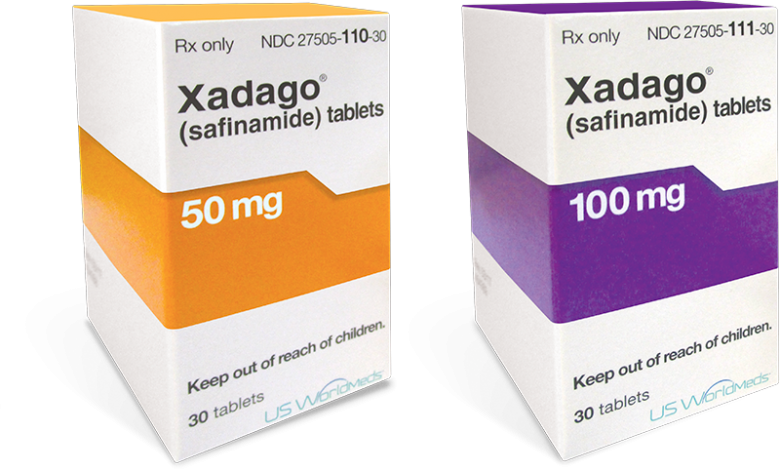
XADAGO is available
in 2 dosages strengths1
The recommended starting dose is 50 mg once daily
After 2 weeks, daily dosage may be increased to 100 mg once daily on the basis of individual clinical need and tolerability
- Can be used with selective serotonin reuptake inhibitors (SSRIs) at the lowest effective SSRI dose
- If a dose is missed, the next dose should be taken at the same time the next day
- XADAGO 100 mg/day should be tapered by decreasing the dose to 50 mg/day for one week before stopping
- In patients with moderate hepatic impairment, a lower dosage of XADAGO (50 mg/day) should be used. XADAGO is contraindicated in patients with severe hepatic impairment (Child-Pugh C)